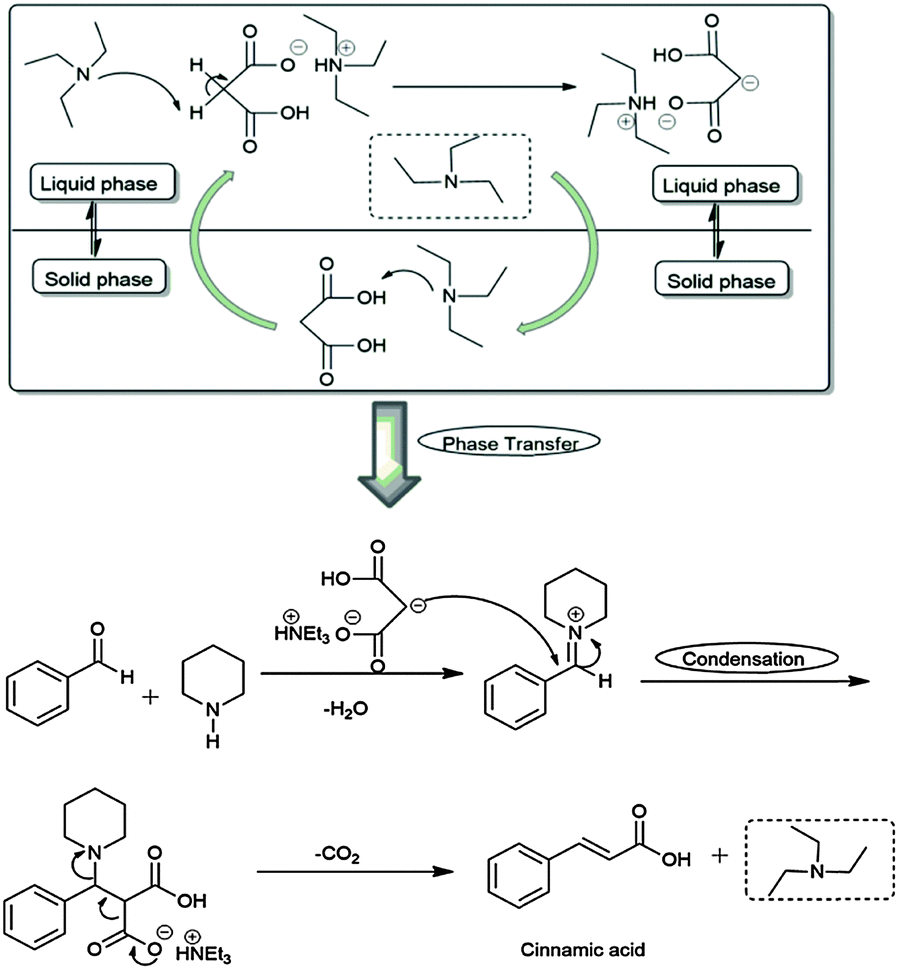
Triethylamine: a potential N-base surrogate for pyridine in Knoevenagel condensation of aromatic aldehydes and malonic acid - New Journal of Chemistry (RSC Publishing) DOI:10.1039/C5NJ03125G
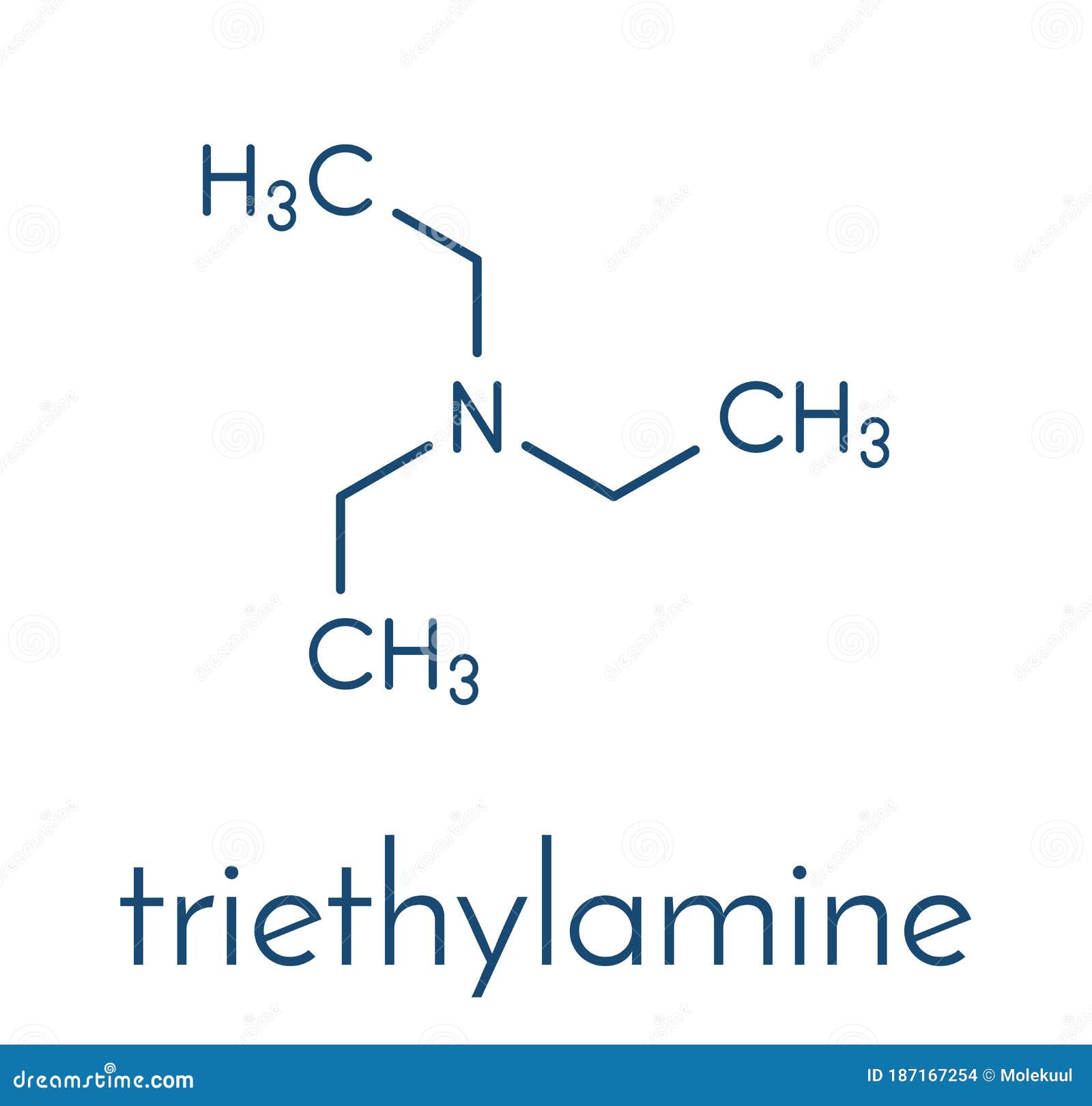
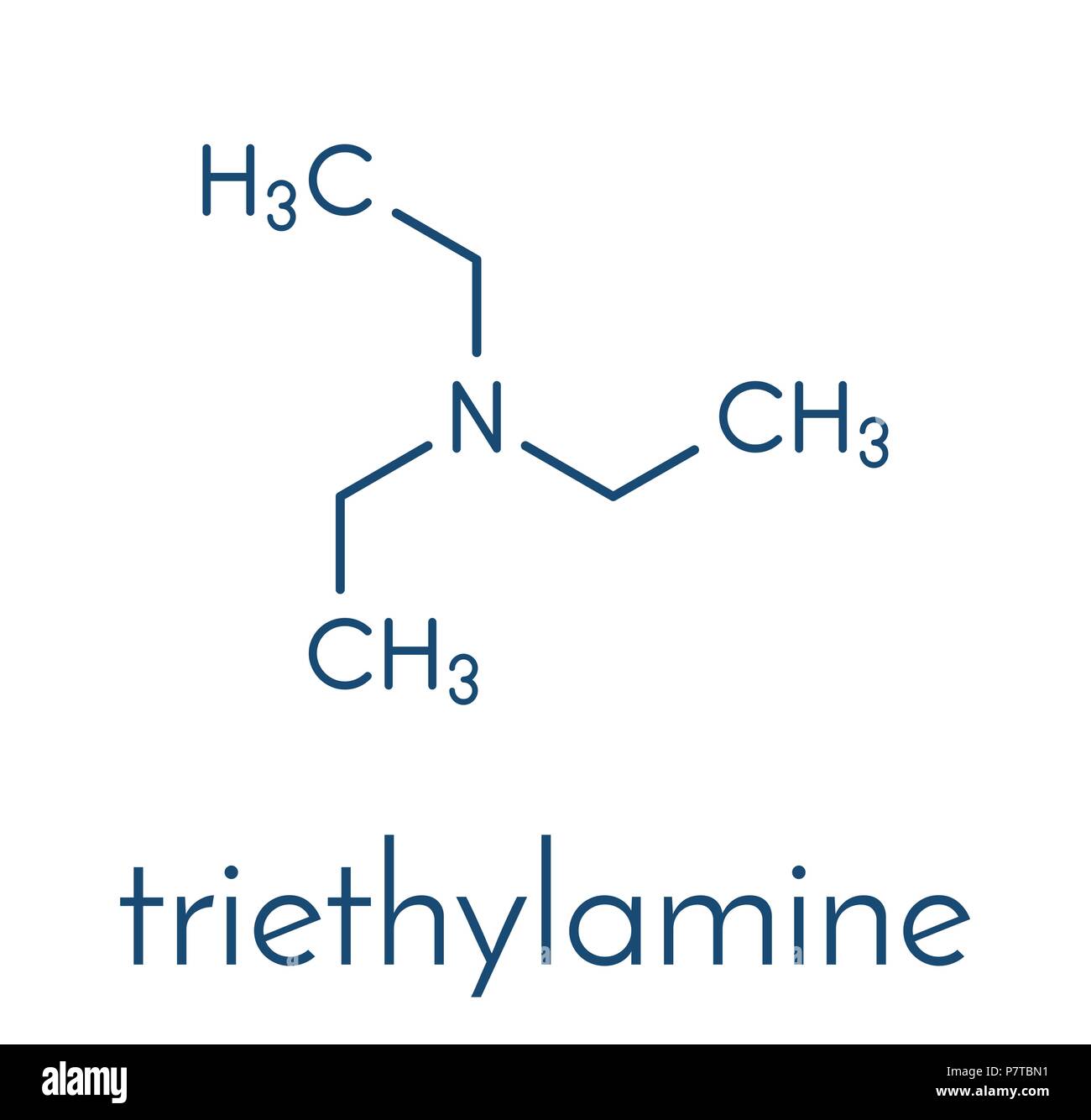
Triethylamine Organic Base Molecule. Skeletal Formula. Stock Vector - Illustration of drawing, triethyl: 187167254
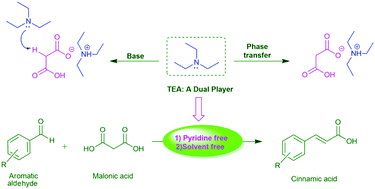
Triethylamine: a potential N-base surrogate for pyridine in Knoevenagel condensation of aromatic aldehydes and malonic acid - New Journal of Chemistry (RSC Publishing)
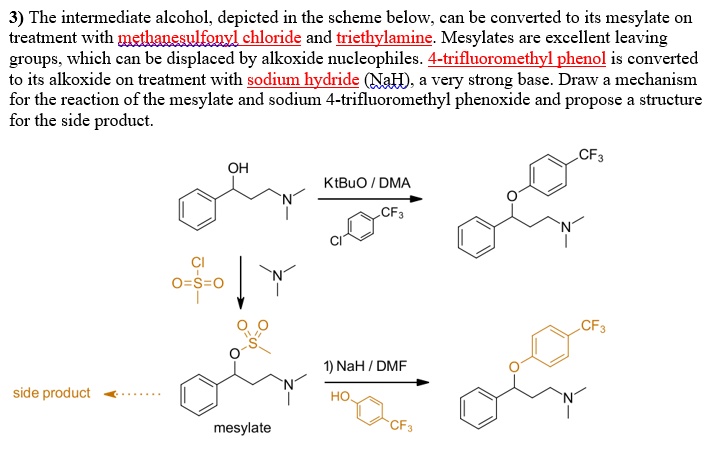
SOLVED: 3) The intermediate alcohol, depicted in the scheme below; can be converted to its mesylate on treatment with methavesulfonvi chloride and triethylamine: Mesylates are excellent leaving groups which can be displaced

PDF) Triethylamine: an efficient N-base catalyst for synthesis of annulated uracil derivativies in aqueous ethanol

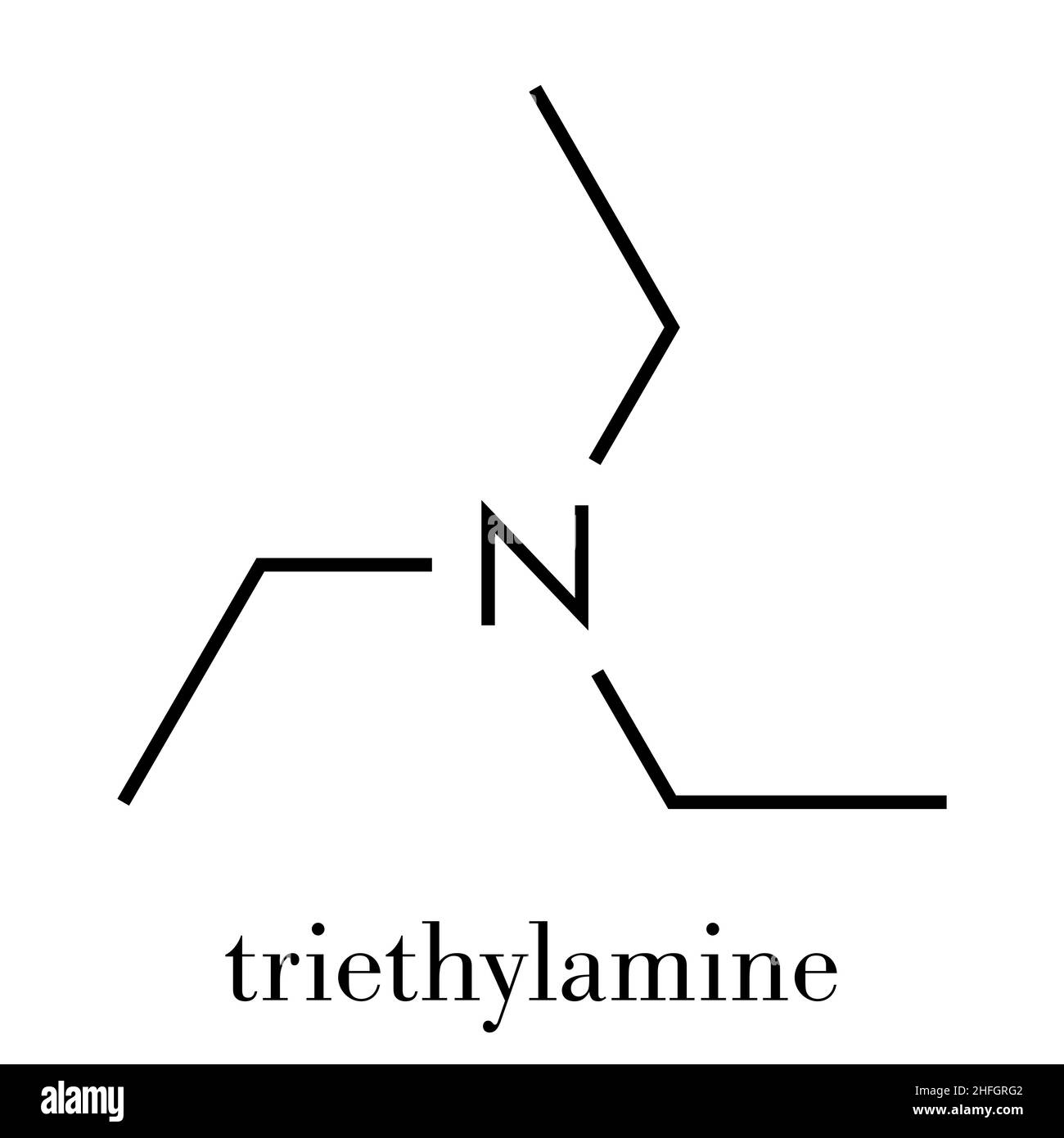
Triethylamine Organic Base Molecule Skeletal Formula Stock Vector (Royalty Free) 1128981863 | Shutterstock

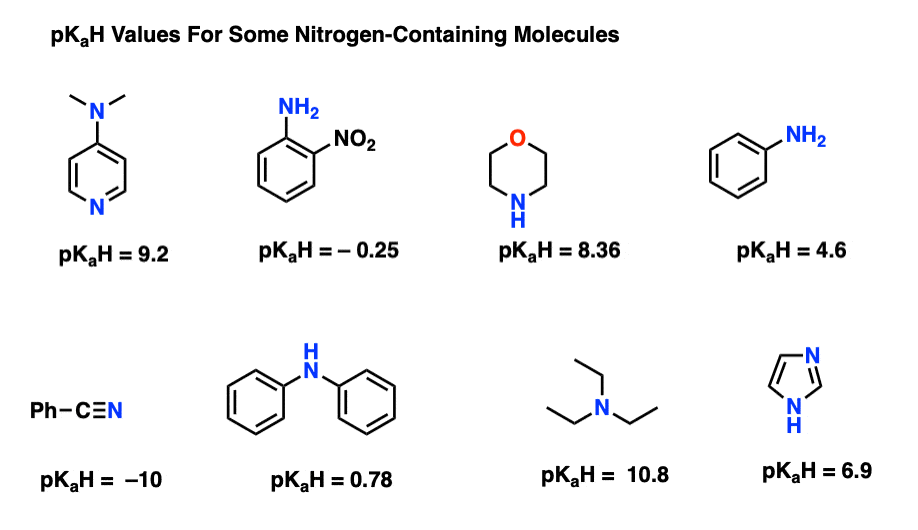
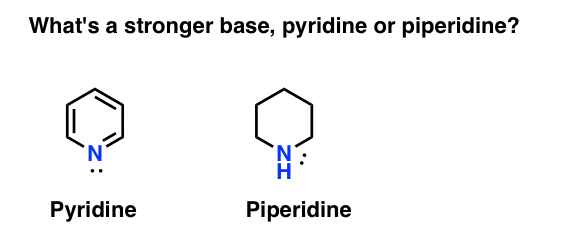
Quinuclidine and triethylamine are both tertiary amines. Quinuclidine, however, is a considerably stronger base than triethylamine. Stated alternatively, the conjugate acid of quinuclidine is a considerably weaker acid than the conjugate acid