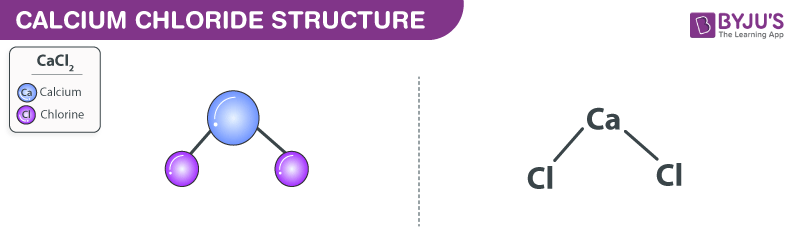
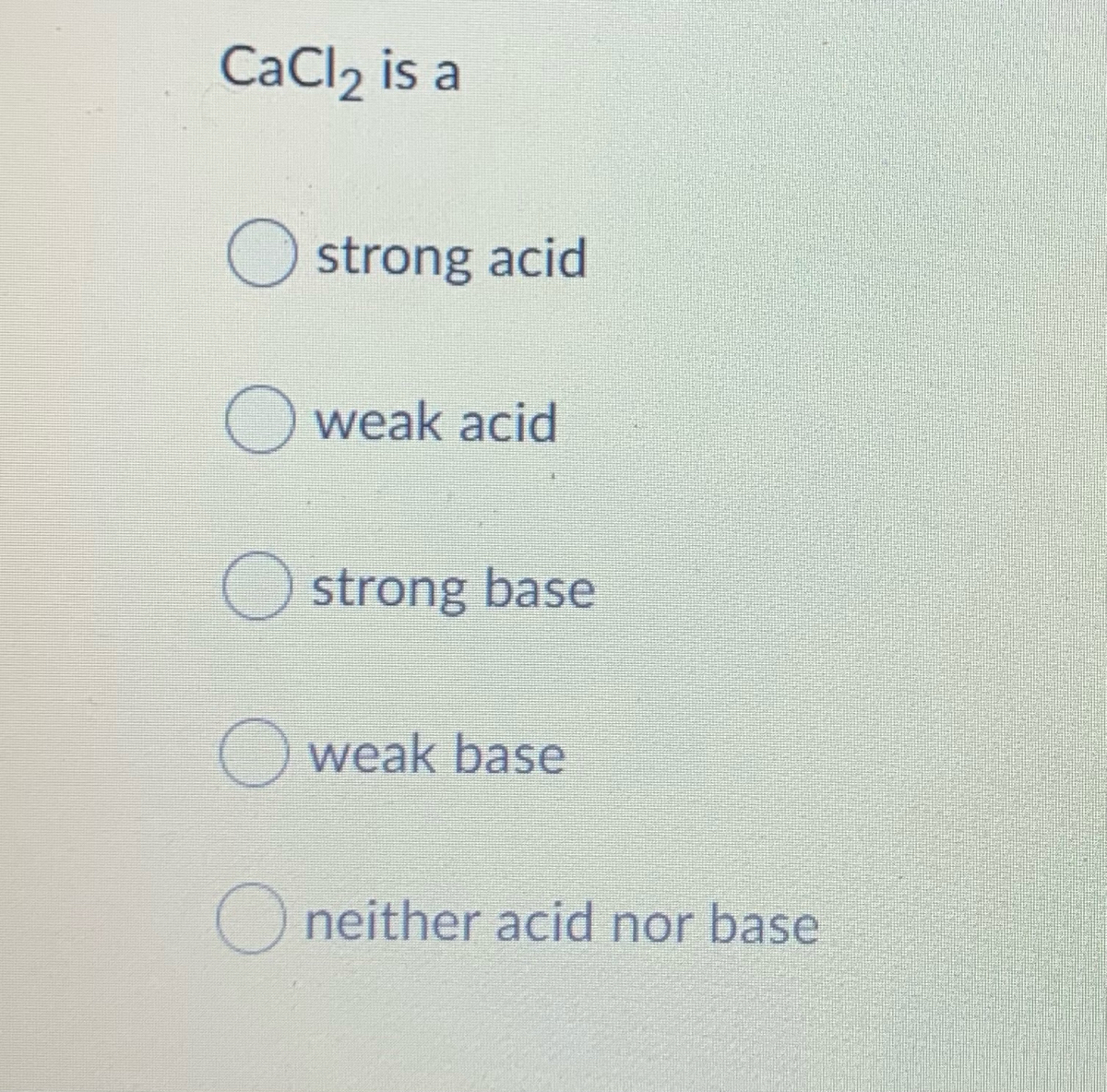
SOLVED: Classify the compounds as acids, bases, or salts. Which are Acids? Which are Bases? Which are Salts? Answer Bank LiCl KOH HBr Ba(OH)2 CaCl2 H2SO3 NH4NO3 H2C2O4

Question Video: Calculating the Mass of Calcium Chloride That Contains a Given Mass of Chlorine | Nagwa

Question Video: Writing a Net Ionic Equation for the Reaction of Solid Calcium Carbonate with a Hydrochloric Acid Solution | Nagwa











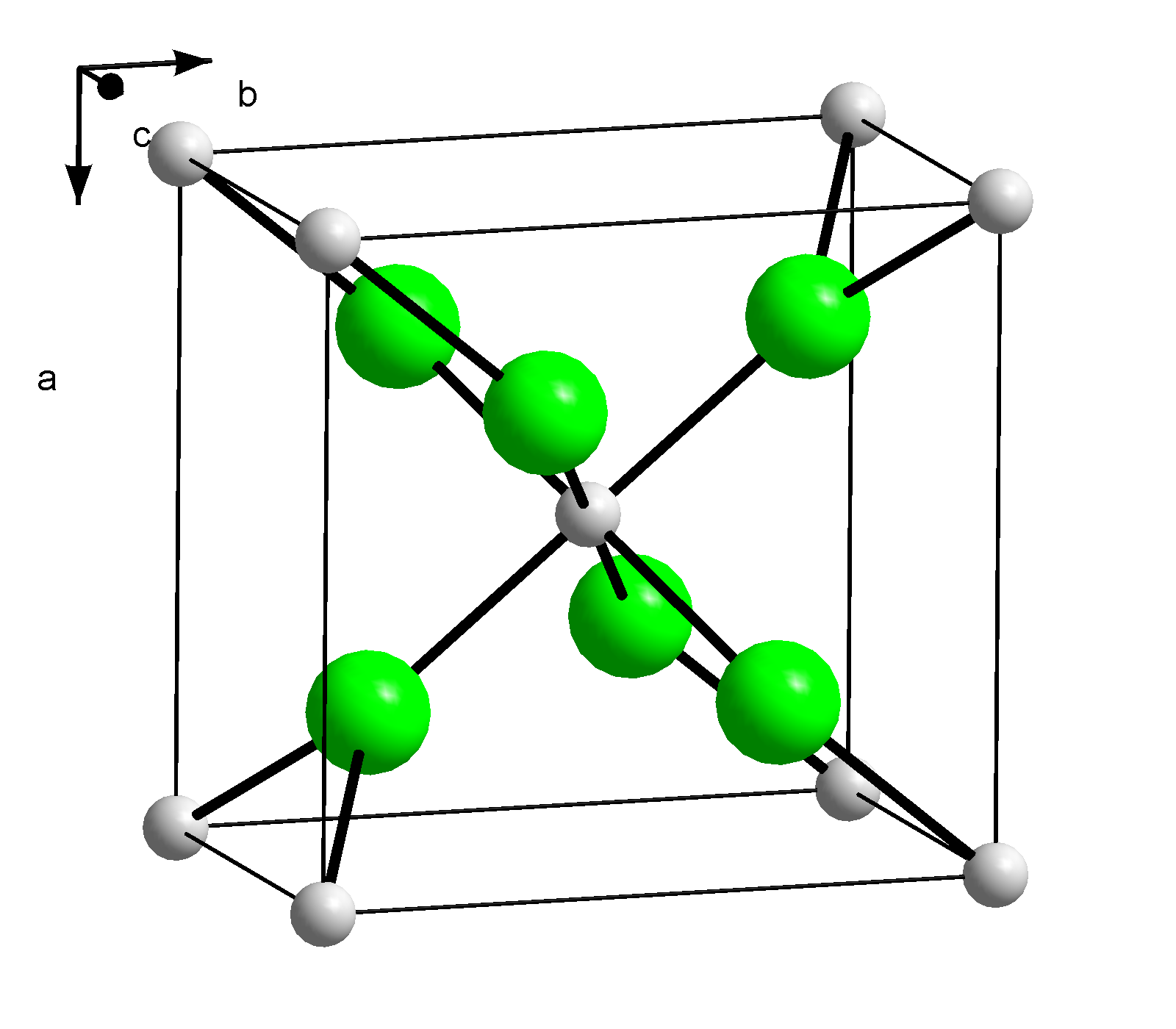
![HCl Gas from conc. HCl(aq) and CaCl2 - [www.rhodium.ws] HCl Gas from conc. HCl(aq) and CaCl2 - [www.rhodium.ws]](https://erowid.org/archive/rhodium/chemistry/equipment/pictures/calcium.hcl.gen.fig.gif)